Research Article - Annals of Experimental Biology ( 2023) Volume 11, Issue 3
Received: 28-Mar-2023, Manuscript No. aeb-23-93336; Editor assigned: 29-Mar-2023, Pre QC No. aeb-23-93336 (PQ); Reviewed: 11-Apr-2023, QC No. aeb-23-93336 (Q); Revised: 15-Apr-2023, Manuscript No. aeb-23-93336 (R); Published: 02-May-2023
Genetic variation is closely correlated with crop improvement. Garlic is mostly propagated through vegetative means; hence, there is a need to induce a significant amount of heterogeneity. Ploidy is induced in vitro by colchicine, which improves allelic variation among individuals. We examined the optimum concentrations of BAP and TDZ for shoot proliferation. The study suggests that BAP (3 mg/L) and TDZ (0.25 mg/L) exhibit considerable growth in shoot regeneration. Twenty-one-day-old calli were subcultured, and the 2nd-generation passage was subjected to a culture medium with BAP, TDZ, and different concentrations of colchicine. The results show that 0.05 mg/L colchicine promotes explant survival. Treatment improved the genetic potential of garlic varieties in vitro but resulted in a lower duplication rate at 0.75 mg/L due to higher toxicity. The LC50 colchicine concentration was 0.03 mg/L-0.05 mg/L. The study suggests that an increase in ploidy level is expected to generate higher molecular diversity in garlic.
BAP, Colchicine, Garlic, Genetic variation, Polyploidy
Garlic (Allium sativum) belongs to the Alliaceae family and is native to central Asia. Garlic has domesticated thousands of years ago and is known to be one of the oldest food crops, as evidenced by fossils [1]. Garlic is made up primarily of water (65%), followed by carbohydrates (26%-30%), followed by other substances such as proteins, lipids, fiber, minerals, saponins, and gallic acid [2]. Other nutrients found in garlic include phenols, the B-complex vitamins thiamine, riboflavin, and niacin, as well as selenium, sulfur, zinc, magnesium, iron, salt, and calcium [3]. They have antibacterial, antifungal, antiviral, antioxidant, and anticancer qualities [4].
Molecular variation is directly proportional to the improvement, but vegetatively propagated garlic has very little variation occurring naturally in garlic due to floral organ abortion and chromosomal aberrations [5]. In vitro propagated plants decrease after continuous propagation. This is because in vitro micropropagation proliferates old cells and forces them to divide into the next generation, which causes a decrease in telomerase activity [6]. Telomerase is a ribonucleoprotein DNA processing enzyme complex that produces telomeres. Due to a decrease in telomerase activity, telomeres become short and potentially delete viable genes. This results in the shortening of plant susceptibility to diseases [7]. This also causes a lowering of molecular diversity and germplasm usefulness.
Genetic enhancement and diversity can be improved with biotechnological approaches using mutagens, and colchicine is the most widely used in inducing polyploidy levels in many garlic varieties [8,9]. Colchicine blocks mitotic chromosomal doubling in metaphase by attaching to the pointed-end microfilaments and the end of the microtubule, disrupting microfilaments an d microtubule polymerization [10]. Due to the high cell/cytoplasm ratio, polyploid plants have a larger clove site and morphology than diploid plants. Polyploidy plants induced by colchicine in vitro result in an increase in DNA content and affect epidermal surface cells [11]. Polyploidy has a thicker cell size but a low cell number per leaf blade compared to diploids [9].
Although colchicine induces polyploidy and enhances genetic diversity, the concentration of colchicine is crucial for the success of ploidy induction. This concentration depends on bulb size, shape, age of the plant, passage, and growth stage [12]. Colchicine treatment of 0.5% for 12 hours and 24 hours resulted in a negative response, i.e., A low number of saplings stunted root growth, thicker stems, and physiological responses such as enlargement of stomata, lower number of stomata and increased chromosome number compared to the control. The degree of plant sensitivity against colchicine was studied based on the Lethal Concentration (LC). The optimum concentration to produce the most mutants was mainly around the lethal concentration of 50 [13].
Initiation and Regeneration of Garlic (Allium sativium L.)
Garlic cloves were selected as explants for micropropagation studies, and 20 accessions were collected from MGM-KVK, Aurangabad. belong to the variety “Godavari” (G1) developed by the NHRDF, which was characterized as a diploid and late mature variety in garlic [1,14,15]. Explants were pretreated with 1% bavistin for 30 minutes to remove fungal contamination, surface sterilized with mild alkaline salts 1% sodium hypochlorite and 2% hydrogen peroxide for 10 minutes each [16,17]. Then rinsed with 70% ethanol followed by rinsing 3 times with ddH2 O and subsequently inoculated in MS medium, 2% sugar, 1 mg pyridoxine-HCl, and MSvitamins at different concentrations of BAP and TDZ [18,19] (Table 1). The pH of the medium was adjusted to 5.8. The sterile cultures were transferred to a fresh medium and maintained for 3 weeks at 24°C /26°C under a 16/8 hours photoperiod using fluorescent light (45 μmol/ms) [17,20].
| Sr. No | Symbol | Treatment 1 (BAP) mg/L | Treatment 2 (TDZ) mg/L |
|---|---|---|---|
| 1 | T1 | 0.5 | 0.25 |
| 2 | T2 | 1 | 0.25 |
| 3 | T3 | 1.5 | 0.25 |
| 4 | T4 | 2 | 0.25 |
| 5 | T5 | 2.5 | 0.25 |
| 6 | T6 | 3 | 0.25 |
| 7 | T7 | 3.5 | 0.25 |
Polyploidy induction in vitro with colchicine
The polyploidy induction experiment was conducted with a completely randomized plan, with six treatments and five replications of each [21]. Twenty-one-day-old seedlings were subcultured, and the 2nd-generation passage was subjected to in vitro culture medium [15, 22]. In each replication, one bottle containing approximately 4 calli (total Population 140) was subjected to six concentrations with five replications of colchicine 0.00 mg/L, 0.02 mg/L, 0.04 mg/L, 0.06 mg/L, 0.08 mg/L, and 0.1 mg/L with charcoal and 3 mg/L BAP, 0.5 mg/L TDZ, 3% sugar, and 0.8% agar‒agar[23]. Table 2 cultures will be maintained at 2500 Lux light density with a photoperiod of 16 hours of light and 8 hours of dark provided by cool white fluorescence. Additionally, for better shoot initiation, the culture was kept in the dark at 25°C (± 2°C) and 70% relative humidity [22]. Biometric observations were conducted every 7 days intervals for evaluation of shoot, root, leaf, and explant size [24].
| Sr. No | Symbol | Treatment 1 (BAP) mg/L | Treatment 2 (TDZ) mg/L | Treatment 3 Colchicine |
|---|---|---|---|---|
| 1 | T1 | 3 | 0.25 | 0 |
| 2 | T2 | 3 | 0.25 | 0.025 |
| 3 | T3 | 3 | 0.25 | 0.05 |
| 4 | T4 | 3 | 0.25 | 0.075 |
| 5 | T5 | 3 | 0.25 | 0.1 |
| 6 | T6 | 3 | 0.25 | 0.125 |
Biometric observation
• Several shoots: The number of shoots that proliferated was measured after inoculation and recorded. The shoot length was measured from the base to the tip of the plantlet at the time of subculture, and the average length was expressed in centimeters (cm).
• Several leaves: The number of leaves that proliferated was measured after inoculation and recorded.
Characterization of polyploidy by cytological and molecular approaches
• G-banding Chromosome Counting: Four-day-old root tips (1 cm-2 cm approx.) of treated plants were taken and kept under submerged conditions in 1% trypsin for 1 hour, after which the root tips were washed three times and immediately placed in absolute methanol for fixation [25]. Slides were air-dried at room temperature for 1 h. After that, 1 drop-2 drops of diluted Giesma-Stain (1:3) for 1 hour were removed from the stain and washed 3 times [25,26]. Root tips were dissected (2 mm) and placed on a glass slide. Then, 1 drop of cedar wood oil was added to increase the refractive index and visualized under the microscope at 100X, 400X, and 1000X magnification [22,27].
• DNA extraction and quantification: Total genomic DNA was isolated from the young leaves of the treated garlic plantlets. Young tissue (0.2 gram) was subjected to extraction using CTAB extraction buffer, incubated for 40 minutes at 60°C, and then centrifuged for 10 minutes at 14000 rpm [28]. The supernatant was collected, and an equal amount of (24:1) chloroform:isoamyl alcohol was added and mixed with gentle shaking [29]. Tubes were again centrifuged at 14000 rpm for 10 minutes, and layers were separated (aqueous phase, interphase and organic phase). The aqueous phase containing DNA was collected and transferred to a fresh tube, and an equal amount of chilled absolute ethanol was added. Centrifugation was carried out at 10000 rpm for 5 minutes at 10°C. Ethanol was discarded [30]. The DNA pellet was air-dried and later dissolved in 50 µL of TE buffer. The DNA samples were quantified using a nanodrop. The concentration (ng/ml) and absorbance at 260 nm/280 nm were observed [31,32].
Statistical analysis
The data obtained on various observations were analyzed by the “analysis of variance” method (Panse and Sukhatme 1967)
In vitro regeneration
Surface sterilization treatments were very effective, and almost 80% of the explants entered the shoot proliferation cycle (Figure 1). MS medium supplemented with BAP and TDZ with 6 replications, developed for garlic proliferation, efficiently induced shoots, and explants were inoculated on MS medium with cytokines with auxin. Explants cultured on MS basal medium without any cytokines took more time to sprout (18 days) and had low efficiency (30%). However, the explants sprouted within 10 days of inoculation on MS medium supplemented with BAP and TDZ (Table 1).
Multiplication
All the results with six treatments and 5 replications showed positive results on shoot proliferation and multiplication in biometric observation of shoots and leaves per explant at 15 days, showing the effect of BAP and TDZ (Tables 2 and 3). Treatment 6 is significant. Shoot initiation of explant 13.8 and nonsignificant treatment 2 (4.8) an average of treatment (T1, T2, T3, T4, T5, T6) is 9.71. The data presented in Table 4 and Figures 2 and 3 show that the average number of leaves of explants, i.e., the results shown in Table 4 and Figures 2 and 3 were significant per treatment (T1, T5, T6), while the treatments (T2, T3, T4) were similar.
| Treatment | Symbol | Day of Initiation | No of leaves per explants | Height of explants(mm) |
|---|---|---|---|---|
| Treatment 1 | T1 | 21.6 | 3 | 57.2 |
| Treatment 2 | T2 | 19 | 2 | 35.6 |
| Treatment 3 | T3 | 18.5 | 1 | 43.6 |
| Treatment 4 | T4 | 17.5 | 2 | 50.6 |
| Treatment 5 | T5 | 17.3 | 1 | 47.6 |
| Treatment 6 | T6 | 15.5 | 2 | 57.3 |
| Treatment 7 | T7 | 20.5 | 3 | 57.5 |
| CV @1% | 9.75 | 26.27 | 5.87 | |
| CD(0.01) | 2.08 | 0.85 | 2.25 |
| Treatment | Symbol | No of shoot per explant | No of leaves per explant | Height of Explant (mm) |
|---|---|---|---|---|
| Treatment 1 | 9.16 | 9.16 | 2.3 | 94 |
| Treatment 2 | 4.86 | 4.86 | 2 | 70.5 |
| Treatment 3 | 8.74 | 8.74 | 2 | 83.5 |
| Treatment 4 | 9.6 | 9.6 | 2 | 108.6 |
| Treatment 5 | 10.6 | 10.6 | 2 | 98.8 |
| Treatment 6 | 10.84 | 10.84 | 2.3 | 113.5 |
| Treatment 7 | 12.94 | 12.94 | 2.6 | 111.3 |
| CV (@ 1%) | 9.04 | 29.8 | 5.847 | |
| CD (0.01) | 2.26 | 0.85 | 1.52 | |
Polyploidization
All garlic tissues treated with colchicine were expressed and showed lower regeneration potential, and most of the plants were yellow and finally died. The survival value ranges between 40%-45% and is much more affected by colchicine time and concentration (Table 5). With increasing colchicine concentration, the survival rate is inversely proportional. The highest lethality was observed at 0.1 mg/L for 48 hours. At 21 days after colchicine treatment, most surviving plants showed normal growth, and most of the plants showed root initiation. On average, control plants produced 8 shoots, 9 shoots, 12 roots, 15 roots, and 2 leaves, 3 leaves. However, surprisingly, treated and surviving plants showed similar numbers of shoots and roots after colchicine exposure. The highest number of shoots and roots was calculated for plants regenerated from tissue treated with 0.06 mg/L and 0.08 mg/L (Table 6 and Figures 3 and 4).
| Sr. No | Treatment | No of Shoot | Shoot Length | No of Leaves | No of Root | Root Length |
|---|---|---|---|---|---|---|
| 1 | Treatment 1 | 9 | 57.2 | 2 | 3 | 16.52 |
| 2 | Treatment 2 | 4.8 | 35.6 | 2 | 2 | 15.6 |
| 3 | Treatment 3 | 4.6 | 43.6 | 1 | 2 | 13.6 |
| 4 | Treatment 4 | 4 | 50.6 | 2 | 2 | 10.6 |
| 5 | Treatment 5 | 2.6 | 47.6 | 1 | 2 | 4.6 |
| 6 | Treatment 6 | 2 | 57.3 | 2 | 0 | 0 |
| Sr. No | Treatment | Diploid | Triploid | Tetraploid |
|---|---|---|---|---|
| 1 | Treatment 1 | 4 | 0 | 0 |
| 2 | Treatment 2 | 2 | 0 | 1 |
| 3 | Treatment 3 | 1 | 2 | 0 |
| 4 | Treatment 4 | 0 | 2 | 1 |
| 5 | Treatment 5 | 0 | 0 | 2 |
| 6 | Treatment 6 | 0 | 1 | 1 |
Chromosome counting
Chromosome counting of tissue culture-raised plants after colchicine treatment was examined using the G-banding chromosomal counting method, and the results showed that out of 30 treated plants, only 17 (56.6%) survived out of 16 surviving diploid (2n=16) 7 (17.5) tetraploid (2n=4x =32) and very few showed triploids (2n=3x=24).Table 6 presents the chromosome counting results.
Photometric analysis
Photometric analysis of ploidy level using Nanodrop absorbance at 260 nm/280 nm. The data show that three different ploidy levels are diploid, triploid, and mixoploid. The mean DNA content found in colchicine-treated plants was 8845 ng/µl, which was greater than that in the control (3658 ng/µl), as shown in Table 7 and Figure 5. The samples were diploid, triploid, and mixoploid. The optical density of total DNA content in Allium spp plants treated with 0.3% colchicine for 18 hours was approximately one and a half times that of diploid DNA, confirming triploids. The total DNA content of 0.4% treated plants after 18 hours was nearly twice that of the tetraploid control groups. Interestingly, plants treated with 0.1% colchicine had three times that of control groups, indicating that they were mixoploids, as there were no significant differences when compared to diploid control plants.
| Sr. No | Sample ID | A260/A280 | A260/A230 | DNA Content (ng/ml) |
|---|---|---|---|---|
| 1 | GRSC1 | 1.77 | 1.03 | 4586.46 |
| 2 | GRSC2 | 1.86 | 1.11 | 4569.56 |
| 3 | GRSC3 | 1.9 | 1.33 | 7548.58 |
| 4 | GRSC4 | 1.69 | 0.94 | 7549.58 |
| 5 | GRSC5 | 1.77 | 1.05 | 9897.5 |
| 6 | GRSC6 | 1.46 | 0.84 | 7556.44 |
| 7 | GRSC7 | 1.88 | 1.14 | 4569.36 |
LC50 analysis
Higher concentrations of colchicine affect callus color and quality, and low concentrations of colchicine do not show any kind of change compared to the control (Table 8). The results showed that 0.05 mg/L colchicine promoted explant survival. Treatment improved the genetic potential of garlic varieties in vitro but resulted in a lower duplication rate at 0.75% due to higher toxicity. The LC50 colchicine concentration was 0.03 mg/L-0.05 mg/L (Figure 6 and).
| Lethal Concentration | Model | Coefficient | Concentration |
|---|---|---|---|
| 0.10 | 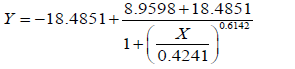 |
A= 8.9598 | 0.4241 |
| B=18.4851 | |||
| C=0.6142 | |||
| LC50 Analysis | |||
The experimental findings of this study are presented with the above heading and are discussed in the following sections. Although it originated in west China and northeastern Asia, garlic is a major plant in Maharashtra and Karnataka due to the Indian subcontinent’s favorable climate [14]. Because of the tiny bulb size and constrained production area of low-yielding types, there is a substantial disparity between the demand and supply of garlic, according to recent studies [16,33].
Mean performance of in vitro regeneration micropropagation study
Studies evidenced by researchers suggest that at low quantities, TDZ causes de novo to shoot organogenesis and somatic embryogenesis at higher degrees of exposure. To detect metabolites in control and TDZ-treated tissues, researchers used an untargeted metabolomics technique. TDZ-induced alterations in metabolism were discovered using statistical methods such as metabolite clustering, pattern and pathway tools, logical algorithms, and synthetic biotransformation. The effect of BAP on in vitro regeneration is evidenced in recent publications [20,34]. It suggests that if a certain concentration of BAP and IAA is used, the combination can be successfully used in tissue culture. Auxin combined with cytokinin (BAP) at the optimum concentration can stimulate explant growth, particularly in the creation of leaves, shoots, and intensive sections. The tissue can produce a variety of organs (leaves or flowers). SAM synthesis is a complex process that is controlled by hormones found in plant tissue. The hormone cytokinin influences SAM growth to create new shoots. In response to environmental cues, cytokinins participate in cell creation, bud expansion, chloroplast formation, and plant metabolism [35]. From this perspective, this study aimed to investigate the effects of growth hormones, colchicine concentration, and immersion time on the ploidy level of garlic to understand genetic variability [22]. For increased cell and shoot proliferation from the callus, garlic in vitro culture can be induced in a medium supplemented with various concentrations of TDZ and BAP (benzyl aminopurine) [18,19]. The results indicate that the TDZ and BAP combination was effective in initiating rapid shoots. In comparison to all other treatments, T1, T6, and T7 exhibited significantly longer shoots than T2, T3, and T5. The above results were taken into consideration for further studies, and 21 day old plantlets were subcultured in new medium with T6 supplemented with colchicine. For instance, colchicine induces meristematic basal discs of garlic plants in vitro at concentrations of 0.25 mg-0.5 mg and immersion times of 24 hours, 36 hours, and 48 hours [20].
Colchicine treatment and ploidy induction
The potency of colchicine for ploidy induction was performed at different concentrations and immersion times, as presented in the previous chapter, which shows that the highest lethality is at 0.1 mg/L and the highest level of ploidy is observed at 0.05 mg/L-0.06 mg/L. This finding reveals the potency of colchicine in ploidy induction. However, for a long time, colchicine has been an important tool in the induction of ploidy, and it has also been noted that the concentration of colchicine is crucial for ploidy induction [8, 13]. Scientists have studied various concentrations to optimize the effective concentration for significant results [7]. Colchicine at high concentrations for extended periods may reduce survival, whereas colchicine at low concentrations for shorter periods may promote survival. The application of colchicine at a 0.5 mg treatment improved the genetic potential of garlic varieties in vitro but resulted in a lower duplication rate of 0.75% due to higher toxicity [16]. The application of colchicine increased the ploidy level, and an increase in ploidy is expected to result in larger bulb size. Larger tuber size is expected to increase the overall tuber weight and total garlic production [36].
Study of performance of colchicine at lethal concentration (LC50) value
Results were compared with Ahmed and Bridge was found significantly at par used colchicine solution of 0.05% concentration for a short duration (1 hours, 2 hours, 4 hours and 8 hours) [16, 26,33]. Simultaneously, 4 different concentrations of colchicine (10 mgL-1 , 20 mgL-1, 30 mgL-1, and 40 mgL-1) at longer durations of 3 days, 6 days and 9 days. The LC50 colchicine concentration was 0.03 mg/L- 0.05 mg/L.
The highest size (4.0 cm) and weight (5.4 grams) of calli were found in 10 mgL-1 colchicine treatment for 3 days, which is closely related to our study.
[Google Scholar] [Cross Ref].