Research Article - Annals of Biological Research ( 2017) Volume 8, Issue 1
Hidol (Shidol), Hukoti, Namsing, Ngari are the four different types of traditional fish products popular in Assam as well as other parts of North East India. Of these products, hidol is largely and exclusively used by the people of different communities of Assam because of its unique flavor and aroma. Present investigation was intended to study on hidol through comparative analysis on extraction and quantitative estimation of proteins extracted from both the fermented ie hidol and unfermented fresh fishes. Experimental study involved protein extraction from both hidol and fresh (unfermented) fish by using alcalase enzyme and quantitative estimation following the methods as described by Lowery et al. (1951). The protein quantity of the hidol and unfermented fresh fish is found to be 363.798 μg/ml and 677 μg/ml respectively.
Fermented fish, Hidol, Alcalase, Protein, Quantitative estimation, Assam
Hidol or Shidol is one of the popularly known traditional fermented fish product widely used by the people of different ethnic communities of Assam and various parts of North East India that play an important role in providing the nutrition of the people. Fermentation is one of the oldest and most economic methods for production and preservation of foods. In addition to preservation, fermentation provides basic component of the diet that enhances flavor, increases digestibility, therapeutic value and taste of the product. The North East India possesses rich diversity of different species of fishes. Apart from the river Brahmaputra and its tributaries, the fish habitats of this part of India are also enriched by water bodies like beel, swampy areas, lakes and a number of streams. Puntius sophore (puthi), Puntius ticto, Setipinna phasa (gangetic hairfin) are the common fish species used for preparation of hidol in thye study area. Hidol is most common and popular fermented fish product used by the people of entire North East India as well as in West Bengal due to its flavor and aroma. It has several local names like hukoti by Sonowal Kochari and Ahom community, Namsing by Mising community of upper Assam and Ngari by the people of Manipur that has been associated with socio economic life of the said communities. The preparation and fermentation process of all these products are almost same expecpt little differences in the fermentation time and some additive ingredients which are locally used by different communities. Our present work is based on hidol which are commonly found in the markets of upper Assam exclusively prepared from Puntius spp. It is mainly used as an ingredient for the preparation of different traditional curries that acts as appetizer. The aim of the present work is based on protein content of the fermented fish product and for locating the emphasis of fermentation on fish protein. The experimental study involved the enzymatic extraction and evaluation of the protein content from both the fermented product and fresh (unfermented) fish Puntius sophore [1-5].
Traditional method for preparation of hidol
The freshly collected Puntius sophore (puthi) are sundried or dried over kitchen fire for a few days (around 5-7 days) until it is too hard to be handled without breaking the flesh of the fish. After that it is filled in airtight earthen pot (koloh) of volume around 4-10 kg capacity. Alternatively, mature bamboo cylinder approximately 1.5 -2 feet long containing node on one side and sealed another side is made airtight by dried leaves of banana also used for fermentation. Before inserting into the pot, the raw material is also soaked with oil extracted from vegetables or some other fishes. The filled in earthen pots or bamboo cylinders are again sealed airtight to provide anaerobic condition for proper fermentation. Then it is stored at room temperature for several weeks for fermentation. Earthen pot is also buried under soil by some communities for several weeks for fermentation.
The study was conducted at Namrup (27.1836° N, 95.3469° E) that falls under Dibrugarh district of upper Assam. The samples were collected in sterile container from the dry fish retailer (who collected the material from nearby villages of Namrup) of the Namrup daily market. They were brought to the laboratory and stored at 4ₒ C in a refrigerator. Knowledge about the preparation, additives and related data were gathered from the fish retailer as well as from the people of nearby villages (Figures 1 and 2).
Chemicals and enzymes
The chemicals and enzyme used in the study include potassium dihydrogen phosphate, dipotassium hydrogen phosphate, concentrated sulfuric acid, concentrated hydrochloric acid, bovine serum albumin as a standard protein, copper sulfate, alkaline sodium carbonate, sodium hydroxide, Sodium potassium tartarate, 2 N Folin Ciocalteu’s phenol reagent and alcalase enzyme (Sigma ctg. no-30079, Sigma Aldrich, Natherland).
Experimental procedure
Determination of pH: 5 gram of sample was homogenized with 10 ml of distill water and pH was measured by using a standard pH meter.
Determination of moisture content: Moisture content was determined by oven drying methods. 5 gm of each sample was taken on a separate aluminum plates and total weight with the sample was taken. After that plate with the sample was put in a controlled hot air oven and was dried at 105°C till the constant weight was achieved [6-11].
% of moisture=(weight of the sample – weight of the dried sample)/weight of the sample × 100
Determination of ash content: Ash content was determined by AMC method. A porcelain dish was heated in a furnace at 550-600°C for 20 minutes, then removed and cooled in a desecrator. 5 gm of sample was taken and spread it out evenly on the dish. Weight of the dish was accurately taken in a electronic balance ( Model-HK-JC 220AB, Galaxy Scientific India) before and after sample was placed. 1 ml of magnesium acetate solution (25 g of anhydrous magnesium acetate made up to 100 ml in water) was evenly distributed over the sample. At first initial heating of the dish was done in a burner for drying and charring of the sample. After that the dish was placed in the furnace and heated at 550-600°C for 3 hours. Cooling was done in a desecrator and again reheating was done for 30 minutes, this process of cooling heating and reweighing was continued until successive weighing was agreed.
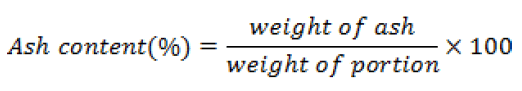
Enzymatic extraction of protein
The enzymatic extraction of the protein from the fermented sample (hidol) and fresh fish (Puntius sophore) was carried out separately. Preparation of each raw material was slightly different from each other but the entire protocol used for the purpose was same. Approximately 10 grams of fresh Puntius sophore was brought to the laboratory and washed with tap water and minced. 10 grams of the sample was weighed, transferred to a glass bottle and placed in a water bath at 90°C for 10 minutes to deactivate the endogenous enzymes. Then the tissue was homogenized by a homogenizer (Teflon pestle tissue homogenizer, JSGW, 633/1) by adding 50 ml of potassium phosphate buffer. The pH of the mixture was adjusted to 7.5 by adding 1 N NaOH. The temperature of the mixture was again optimized at 55°C by placing the glass bottle containing the mixture on a water bath shaker of 140 rpm. After that 0.5% alcalase (ie.0.05 gm in respect to 10 gm of raw sample) was added to the mixture and kept for 1 hour for enzymatic hydrolysis of protein. The mixture was again placed in another water bath operating at 90°C for 5 minutes to inactivate the alcalase enzyme.
After proper enzymatic hydrolysis of protein the components of the mixture were separated from each other by a centrifuge (Model CM12, Sr. No VCEJ- 6885, Remi Electronic Ltd. India). The centrifugation of the mixture at 4100 rpm for 40 minutes had separated the mixture into four layers - upper oil layer, light lipid layer, a soluble clear protein layer, and bottom sludge layer containing the remaining fish tissue. The upper oil layer was carefully removed with the help of a pipette, the middle lipid and soluble protein layer was removed by tiling the tubes and pour it into a filtration funnel without disturbing the sludge. The remaining sludge was discarded. The protein hydrolysate was obtained by filtering the light lipid layer from the soluble protein using Fisher p5 filter paper. The quantitative estimation of protein hydrolysate was done by using the method as described by Lawry et al. (1951).
Determination of protein concentration
The protein concentration was determined by following the method as described by Lawry et al. (1951). The phenolic group of tryptophan and tyrosine residues (amino acids) in a protein produced a blue purple color complex, with a maximum absorption at 660 nm wavelength of light, with Folin-Ciocalteau reagent which consists of sodium tungstate molybdate and phosphate. Thus intensities of the color depend on the amount of these aromatic amino acids. Solution (A) was prepared by mixing 1 gm Na2CO3 and 0.2 gm NaOH in 50 ml of distill water in a conical flask. Solution (B) was prepared by mixing 0.005 gm CuSO4.5H2O and 0.01 g Sodium Potassium Tartrate in 1 ml Distill water (Figures 3-5).
The complex forming solution i.e. working solution was prepared by mixing 1 ml of solution B with 49 ml of solution A. Folin Ciocalteau reagent was prepared freshly by diluting the reagent with distill water in the ration of 1:2.
Bovine Serum Albumin (BSA) was used as the standard protein solution. Several concentrations were prepared for the standard curve. 0.02 g of BSA was added to a 10 ml volumetric flask containing distilled water. The final concentration of the BSA solution was 200 μg/ml. Dilutions ranging from 0 to 200 μg/ml were prepared. The absorbance was taken at 660 nm under UV visible spectrophotometer (Galaxy Scientific India IEU 944/3, New Delhi). Absorbance values of the BSA standards were plotted against the BSA concentrations as shown in Figure 6.
Four types of fish products i.e. hidol, hukoti, namsing, ngari were collected and studied about the sensory quality including appearance, color, texture and flavor. The mean value of moisture and ash content of the four separate samples was presented in the Table 1. Fish protein was extracted from the hidol and unfermented fresh Puntius sophore by taking 10 gm of each. The amount of protein recovered from each sample was calculated out and the result was shown in Table 2. Concentration of the enzymatic extracted protein from whole unfermented 10 gm fresh Puntius sophore was fund to be 677 μg/ml and from the fermented fish product hidol was 363.798 μg/ml. Fermentation involves the breakdown of proteins in the raw fish to simpler substances which are themselves stable at normal temperatures of storage. Breakdown of proteins is brought about by the action of enzymes and sometimes microorganisms are also involved.
| Moisture Content (%) | Ash content (%) | pH | |
|---|---|---|---|
| Ngari | 32.55 | 1.31 | 6.35 |
| Hukoti | 16.67 | 2.01 | 7.56 |
| Namsing | 14.70 | 2.67 | 7.01 |
| Hidol | 29.66 | 1.06 | 6.23 |
| Unfermented Puntius sophore | 36.55 | 2.4 | 5.89 |
Table 1: Quality of hidol.
| Material | Hydrolysis time | Enzyme concentration (%) | Protein yield (%) | Concentration of protein |
|---|---|---|---|---|
| Hidol prepared from Puntius sophore | 1 hour | 0.5 | 35.56 | 363.798 µg/ml |
| Fresh Puntius sophore | 1hour | 0.5 | 53.94 | 677µg/ml |
Table 2: Protein content.
The research was supported by DBT Govt. of India through providing funds as Institutional Biotechnology Hub to Namrup College. The author gratefully acknowledges the co-operation and help received from the fish retailer and people of the different communities of Namrup for providing valuable information regarding preparation and preservation of the sample.