Research - Der Pharmacia Lettre ( 2021) Volume 13, Issue 9
Curcumin is an antidiabetic, anti-bacterial, anti-inflammatory, antioxidant drug having low solubility due to its crystalline nature. Drug is coming under class II BCS classification. Here it is an attempt to improve solubility by inclusion complexation method. For this research work cyclodextrin was used. Cyclodextrin is derived from starch. It of different types alpha, beta and gamma cyclodextrin. Depending upon number of sugar moiety cyclodextrins is classified. β Cyclodextrin used in research work for complexation with Curcumin . X ray Diffraction study was conducted for both pure drug and complex. The pure drug shows sharp peaks in graph which reflects nature of crystallinity. Inclusion complexation was done by Physical mixture and kneading method. Volume, temperature and solvent were optimized. After complexation micromeritics and in vitro dissolution tests were conducted, this gave satisfactory results. Calibration curve was done with distilled water. Phase solubility study was conducted.
FTIR, Phase solubility, BCS, Crystallinity, Amorphous.
Curcumin is a drug used for multiple purpose. It also helps to prevent nerve problem, blindness, kidney damage. It keeps blood sugar low. More than 400 million people across the world under the threat of diabetes [1-4]. It’s having anti-inflammatory effect. The solubility of drug is least in aqueous medium and under BCS class II. Effects of curcumin for diabetic patients are needful and helpful. So combined therapy for diabetes is effective. The study is designed to improve the aqueous solubility by different techniques. Both drugs having challenges for formulations. Solid dispersion can improve the solubility of curcumin [5]. The curcumin can be enhanced its solubility by complexation. It can be formulated by enhancing its dissolution rate. As there as not still studied the combined formulation of complexed formulation with the said techniques, it is important to study the comparative study of drug with complexation and non-complexed active ingredients the successful study may lead to cost effective formulations with an effective formulation for mankind [6].
Materials
The list of apparatus and instruments used in this research are listed below (Table 1).
| SL. No. | Apparatus | Source |
|---|---|---|
| 1 | Analytical balance | Adair Dutta, AD-50 B, Kolkata |
| 2 | Glass wares, beakers, separatory funnel | Borosil |
| 3 | Magnetic stirrer | Remi magnetic stirrer |
| 4 | Syring | Dispovan |
| 5 | Sonicator | Probe Sonicator |
| 6 | UV/VIS. Spectroscopy | Systronic double beam 2203 smart spectrophotometer |
| 7 | Powdered – XRD | Phillip analytical X-ray BV (PW3710) X-ray Diffractometer |
| 8 | Dissolution Apparatus | USP 8 basket Digital Test Apparatus Lab India (Disso-2000) Mumbai |
| 9 | Sieve set | ASTM Standard Sieves, SISSO, India |
| 10 | Distillation apparatus | Borosil |
| 11 | Shaker water bath | Remi shaker water bath |
| 12 | Incubator | Thermoline laboratory incubator |
Micromeritics study-density measurement
Bulk density: First 1 gm drug is weighed accurately and kept in a clean dry graduated measuring cylinder. Then after pouring the dug in to the cylinder, the granular bed made uniform without disturbing much [7]. The volume map measured directly from the granulation. Measured volume is called as bulk volume. The density is called as bulk density.

Tapped density: After measuring bulk volume of the same measuring cylinder is subjected to tap really 200 times by hand. Then volume was detected. This volume is called as tapped volume.
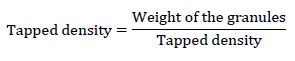
Flow properties
Angle of repose: A glass funnel having tip cut horizontal to the surface was fixed at constant height around 2 cm with the help of a stand and on the tip one graph paper was placed 2 gm of the drug was weighed and directly poured at a time through the funnel [8]. So the granules formed a conical structure having a height. The weight of the firmed core way measured with the help of scale and the perimeter of the core was marked with the help of marker [9]. From this average radius of the formed circle was measured by drawing various diameter through the center, the angle of repose was calculated by the following formula [10].

Where,
ɵ=Angle of repose.
h=height of the formed cone.
r=radius of the circular base on the formed cone (Table 2).
| Angle of repose in degree | Flow property |
|---|---|
| <25 | Excellent |
| 25-30 | Good |
| 30-40 | Passable |
| >40 | Very poor |
Carr’s index: It is one of the most important parameter to characterize the nature of powder and granules (Table 3).
| Carr’s index | Types of flow |
|---|---|
| 5-15 | Excellent flow |
| 12-16 | Good flow |
| 18-21 | Fair to passable flow |
| 23-35 | Poor flow |
| 33-38 | Very poor flow |
| >40 | Extremely poor flow |

Hausner’s ratio: It is an important character to determine the flow property of powder and granules.
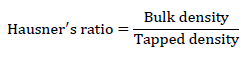
Value less than 1.25 indicates good flow and greater than 1.25 indicates poor flow.
Illuriation on micromeritics study of pure drug
Micromeritics of the study of the pure drugs and their results were reported in the (Table 4).
| Experiment | Result |
|---|---|
| Bulk density | 0.1742 gm/ml |
| Tapped density | 0.2632 gm/ml |
| Carr’s index | 33.82% |
| Hausner’s ratio | 1.51 |
| Angle of repose | 33.52° |
Sieve analysis: An accurate weighed 2 gm quantity of drug was subjected to granulometric study using sieves 22, 30, 44, 60, 80, 100 and 120 using a sieve shaker. Drug is sieved nearly around 10 minutes than the sieves are removed from the sieve shaker and powder retained in each sieves was calculated in percentage form using initial weight taken. The results were given below (Tables 5-7, Figures 1 and 2).
| Sieve no. | Retained amount of drug (in mg) | Percentage retained (%) |
|---|---|---|
| 22 | 0.926 | 46.3 |
| 30 | 0.33 | 16.5 |
| 44 | 0.176 | 8.8 |
| 60 | 0.168 | 8.4 |
| 80 | 0.016 | 0.8 |
| 100 | 0.101 | 5.05 |
| 120 | 0.01 | 0.5 |
| Total | 1.727 | 86.35 |
| Concentration (mcg/ml) | Absorbance |
|---|---|
| 0 | 0 |
| 5 | 0.076 |
| 10 | 0.191 |
| 15 | 0.298 |
| 20 | 0.396 |
| 25 | 0.492 |
| 30 | 0.611 |
| 35 | 0.728 |
| 40 | 0.836 |
| 45 | 0.939 |
| 50 | 1.043 |
| Sl.NO. | Time | %DR |
|---|---|---|
| 1 | 0 | 0 |
| 2 | 2 | 0.76 |
| 3 | 4 | 1.9 |
| 4 | 6 | 2.75 |
| 5 | 8 | 5.63 |
| 6 | 10 | 8.25 |
| 7 | 20 | 15.6 |
| 8 | 30 | 20.18 |
Methodology optimization
Physical mixture method: Drug and beta-cyclodextrin in the ratio 1:2 was taken and were mixed thoroughly with constant trituration, passed through sieve no. 100 and stored in a desicator.
Kneading method: Drug and beta-cyclodextrin in the ratio 1:2 was taken and to it 20 ml methanol was added and were mixed till a thick slurry was obtained with constant trituration, then it was dried at 45°C, passed through sieve no.100 and stored in a desicator (Tables 8-11, Figures 3 and 4).
| Method | % Drug content |
|---|---|
| Physical mixture method | 83.82 |
| Kneading method | 85.62 |
| Sample | Volume of methanol | Percentage |
|---|---|---|
| A | 25 | 55.12 |
| B | 20 | 85.62 |
| C | 15 | 85.05 |
| Parameters | Complex | Pure drug |
|---|---|---|
| Bulk density | 0.294 gm/ml | 0.1742 gm/ml |
| Tapped density | 0.384 gm/ml | 0.2632 gm/ml |
| Compressibility index | 23.43% | 33.82% |
| Angle of repose | 30.068 | 33.52 |
| Hausner’s ratio | 1.306 | 1.51 |
| Time(min) | %DR(PM) | %DR(KM) | Pure Drug(PD) |
|---|---|---|---|
| 0 | 0 | 0 | 0 |
| 2 | 8.3 | 11.58 | 0.76 |
| 4 | 17 | 30.29 | 1.9 |
| 6 | 24.1 | 35.89 | 2.75 |
| 8 | 37.5 | 40.09 | 5.63 |
| 10 | 44.21 | 47.2 | 8.25 |
| 20 | 61.48 | 75.74 | 15.6 |
| 30 | 65.94 | 86.43 | 20.18 |
According to World Health Organization (WHO) reports from 1980 to 2016 the people of diabetes rose from 108 million to 422 million. Between 2000 to 2016 death rates below the age of 18 increased to 5%. Diabetes plays an important role in failure of kidney, blindness, Heart attack. According to WHO diabetes is the seventh leading cause of death. The potent drug curcumin may plays an important role if it will more soluble. The result is satisfactory and it can be formulated in new dosage form along with anti-diabetic drug.